Probing organized lipids in biological tissues using nonlinear microscopy / Cartographier l’orientation des lipides dans des tissus biologiques
[FRA] Une méthode non invasive permettant d’observer in situ comment sont orientés les assemblages de lipides dans les tissus biologiques vient d’être mise au point par les physiciens du Laboratoire d’optique et biosciences (CNRS / Inserm / École polytechnique). Elle ne nécessite ni préparation, ni marqueur. Ces travaux devraient permettre de caractériser certaines pathologies associées à des désorganisations moléculaires au niveau de la peau ou du tissu nerveux. Ils sont publiés en janvier 2013 dans la revue en ligne Physical Review X.
Communiqué de presse CNRS/X. Français English
Probing ordered lipid assemblies with polarized third-harmonic generation microscopy. M. Zimmerley, P. Mahou, D. Débarre, M.-C. Schanne-Klein & E. Beaurepaire. Physical Review X, 3, 011002 (2013). http://dx.doi.org/10.1103/PhysRevX.3.011002.
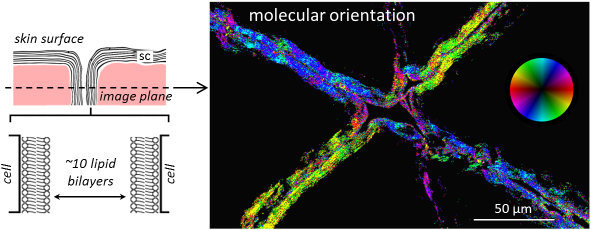
[ENG] Mapping lipid orientation in a human skin sample using nonlinear microscopy. P-THG microscopy can probe molecular order at a micrometer scale in intact, unstained tissues. (Left) Schematic representation of the outermost skin layer. The stratum corneum (sc) consists of flat, dead corneocyte cells surrounded by a multilamellar lipid matrix. This organization is necessary for the skin barrier function. (Right) Map of the average orientation of lipid molecules within sc folds. The image represents a virtual biopsy recorded 30µm below the skin surface. © Ecole Polytechnique, CNRS, INSERM / American Physical Society.
Popular summary:
Bilayer structures formed by ordered assemblies of the chemically ‘‘bigamous’’ lipid molecules (which have chemical affinity for both water and oil) are building blocks found in all mammalian biological cells. In particular, the proper formation of stacked lipid bilayers plays a key role in the function of skin as a protective barrier, and in the electrical connectivity of neurons. Probing and measuring the lipid-molecular ordering in biological tissues, or in situ, and correlating that knowledge with the functions of the tissues is therefore scientifically, and even medically, important. In this study, we establish a novel method for characterizing in situ lipid ordering in human skin samples with subcellular 3D resolution. We do so by exploiting and refining a nonlinear (multiphoton) microscopy technique that depends on third-harmonic generation and on light polarization.
Ordered lipid structures are in fact nonlinear optical materials. When illuminated by an optical (laser) beam, they interact with the incident photons and emit photons with multiples of the energy of the original photons. Third-harmonic generation corresponds to the photon emission of tripled energy or frequency and is the basis of our method. As we have discovered through a theoretical analysis that spans from the molecular scale to the lipid assembly scale, the light resulting from third-harmonic generation in a lipid-bilayer assembly is actually sensitive to how the molecules (or more specifically the chemical bonds in the molecules) are oriented with respect to the polarization of the excitation beam. Since ordering of lipid molecules in a supramolecular assembly is intimately related to their orientations in the assembly, the observable sensitivity to molecular orientation translates into an observable sensitivity to the molecular ordering. Based on this theoretical understanding, we are able to experimentally image, with micrometer resolution, lipid orientation and order not only in artificial lipid assemblies, but also in tissue samples obtained from human skin biopsy. Compared to the existing techniques for characterizing lipid order, such as nuclear magnetic resonance, the optical method represents a huge improvement in spatial resolution, can be used in situ, and has the advantage of not requiring the use of unnatural bulky probes.
Our proof-of-principle study demonstrates the promise of polarization-sensitive third-harmonic-generation microscopy for probing molecular order in biological tissues. We hope that further development of the method, by us and by others, will turn it into a practical method for physiological studies of ordered molecular assemblies.
