Adaptive optics
Adaptive optics and wavefront shaping
Adaptive optics
The aim of adaptive optics for microscopy is to restore a diffraction limited focus within an inhomogeneous sample that introduces wavefront aberrations. This permits improving the contrast and resolution of images recorded deep inside biological samples.
Because most biological tissues are optically inhomogeneous, the quality of focussing within the sample quickly degrades with penetration depth. The resulting spreading of the point-spread function (PSF) affects both the image intensity and the resolution, which cannot be recovered by simply increasing the excitation power.
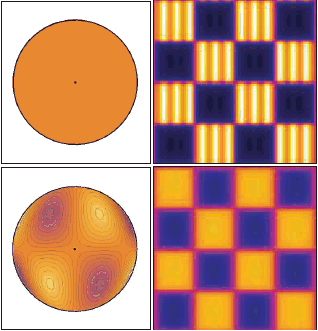
Typical aberrations include spherical aberration (e.g. in the case of a sample whose index of refraction is different from that of the immersion medium), astigmatism and coma (e.g. if the surface of the sample is curved or tilted), etc.
A means of overcoming this limitation is to introduce a wavefront deformation of the excitation beam before the focussing objective that compensates for aberrations created within the sample. This technique, known as adaptive optics in astronomy, can be used in microscopy provided that the aberrations introduced by the sample are known.
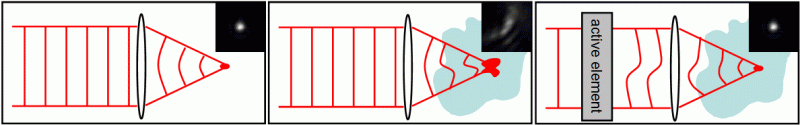
Principle of aberration correction: in the absence of aberration, the focal spot is diffraction limited (left). Aberration introduced by inhomogeneous samples distort the focal spot (center). An active element is used to compensate for these aberrations and restore a diffraction-limited focal spot.
Over the past few years, we have worked on the improvement for nonlinear microscopy of a sensorless method relying on indirect measurement of the aberrations. This method, initially developed in the group of Prof. Tony Wilson at the University of Oxford, relies on the analysis of image quality as a function of applied aberrations to determine the aberration initially introduced by the sample.
Principle of image-based correction. Here a single Zernike mode (astigmatism) is corrected for. A known amount of aberration (bias) is introduced in the chosen mode by use of a deformable mirror, resulting in a change in image quality measured by metric M (here a two-photon fluorescence image is shown and M is the mean intensity). Correction is estimated from at least 3 measurements, and is applied using the same deformable mirror.
A major advantage of this approach is that it does not require a wavefront sensor as the correcting device is also used for sensing.
We have extended this approach to aberration correction in THG microscopy. This technique is highly sensitive to the phase of the excitation beam, so that aberration correction has a dramatic effect on the image quality. Indeed we have shown that correction could improve the THG signal by up to a factor of 2.7 in biological samples.
Combined THG/two-photon fluorescence image of a pollen grain without (left) and with (right) adaptive optics. Purple, THG; Yellow, two-photon fluorescence. Adapted from (Olivier et al., 2009)
By optimizing the correction scheme, we have also reduced the time and illumination required for correction to that of a normal image, so that aberration correction only doubles the illumination time on the sample. As a result we could demonstrate dynamic correction on a developing Drosophila embryo for over 1h.
Drosophila embryo development imaged with THG microscopy before (left) and after (right) aberration correction. Adapted from (Olivier et al., 2009)
The correction can also be updated as a function of depth in the case of thick objects.
3D reconstruction of a 40µm-thick slice in a whole fixed mouse embryo before (left) and after (right) aberration correction. Signal arise from the chromatin (DAPI staining) and from some epithelial cells (cytoplasmic GFP expression). Adapted from (Débarre et al., 2009).
Current topics include the comparison of different aberration schemes and improvement of the correction speed.
RELATED PUBLICATIONS
"3D resolved mapping of optical aberrations in thick samples"
J. Zeng, P. Mahou, M.-C. Schanne-Klein, E. Beaurepaire, and D. Débarre
Biomed. Opt. Express 3(8), 1898-1913 (2012).
"Accuracy of correction in modal sensorless adaptive optics"
A. Facomprez, E. Beaurepaire, and D. Débarre
Opt. Express 20(3), 2837-2849 (2012).
"Simple characterisation of a deformable mirror inside a high numerical aperture microscope using phase diversity"
D. Débarre, T. Vieille and E. Beaurepaire
J. Microsc. 244(2), 136-143 (2011).
"Dynamic aberration correction for multiharmonic microscopy"
N. Olivier, D. Débarre & E. Beaurepaire
Opt. Lett. 34(20), 3145-7 (2009). PDF
"Image-based adaptive optics for two-photon microscopy"
D. Débarre, E. Botcherby, T. Watanabe, S. Srinivas, M. Booth & T. Wilson
Opt. Lett. 34(16), 2495-7 (2009). PDF
