Transfert de ligands étudié par spectroscopie infrarouge
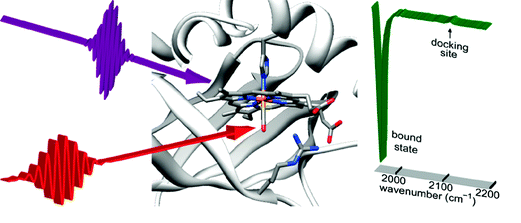
In the case of heme proteins, we explore the vibrational frequency modifications of different ligands (diatomic molecules : CO, NO, O2) in order to probe the ligand evolution during its photo-initiated transfer from one site inside the heme pocket to another. The ligand vibrational frequency depends on its binding site and the associated electron exchange, on the local electric field due to the surrounding amino acids, on the formation of hydrogen bonds with its environment.
Direct observation of ligand transfer and bond formation in cytochrome c oxidase using mid-infrared chirped-pulse upconversion
J. Treuffet, K.J. Kubarych, J.-C. Lambry, E. Pilet, J.-B. Masson, J.-L. Martin, M.H. Vos, M. Joffre, A. Alexandrou
Proc. Natl. Acad. Sc. (USA) 104, 15705 (2007) PDF
CO vibration as a probe of ligand dissociation and transfer in myoglobin
T. Polack, J. P. Ogilvie, S. Franzen, M.H. Vos, M. Joffre, J.-L. Martin, A. Alexandrou
Phys. Rev. Lett. 93, 018102(2004) PDF
